Living systems are not only complex, they are relentless in their demands to be kept alive, with all the attendant limitations and challenges for experimentation and measurement. So-called “cell-free systems” inhabit the space between chemistry and biology proper, obviating some of the requirements of living, cell-based systems and enabling biochemical processes and experimental interventions toxic, contrary, or simply inconvenient to the enterprise of being fully alive. Cell-free systems may deepen our fundamental understanding of life itself, for example, by offering an attractive platform towards building artificial and minimal cells.
Cell-free systems are typically composed from the minimally-prepared extracts of lysed cells or reconstituted from purified biochemical components, supplemented with the desired DNA template and a chemical mixture to supply energy, amino acids, and small molecules to the system.
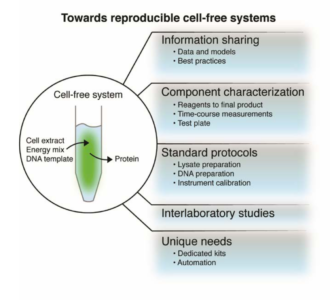
Despite a relaxation of the stringent demands of living, users of cell-free systems must still contend with the inherent complexity of life that is both the compelling strength and confounding liability of cell-free systems. Consequently, a lack of reproducibility persists within and across laboratories engaged in research using cell-free systems.
Complexity in cell-free systems can manifest as variability in function, such as protein production, which generally cannot be designed out or optimized away at the state of the art. Although biological variability undoubtedly contributes to generally poor reproducibility in uses of cell-free systems, this Workshop focused instead on modeling, laboratory practices, and measurement technology to improve reproducibility. Biological variability may then be approached as an enabling feature of engineered biological systems for emerging applications. This view deliberately tempers a pervasive myopic affinity for deterministic systems, which would miss the opportunity to learn control approaches unique to biological systems and harness these for safe and robust applications of engineering and synthetic biology broadly.
Problems with Lysate
The state of lysate preparation for cell-free systems is characterized by non-standardized inputs and irreproducible or non-scalable kits which leads to high expense. Laboratories widely choose to produce their own lysate, even for common organisms, because existing commercial kits are prohibitively expensive, have limited applications, and are not customizable. Commercial kits do not seem to be any more reproducible or characterized than their laboratory-prepared analogs. Consequently, each laboratory typically has one person with so-called “magic hands” who, for undetermined and perhaps indeterminable reasons, is best able to produce suitable lysate for all users and collaborators for that laboratory. Protocols tend to follow historical precedent without optimization, suggesting the possibility that the full range of performance attainable by cell-free systems through optimization of the lysate remains unexplored and potentially considerable.
For example, consider the numerous factors that may introduce variability in uses of E. coli lysates for cell-free systems. The choice of E. coli strain and whether a runoff reaction was performed generally affect the final cell-free reaction. Cell growth is affected by media composition, and the use of defined media could help standardize the preparation of cell lysates. Researchers may find that B strains of E. coli tend to perform better than K strains for no discernible reason. Similarly, researchers may prefer lysate derived from cells grown at nominally different incubation temperatures.
Complexities tend to compound. Lysates may be further affected by the specific starter culture, presence of phage, use of a glycerol stock, use of an ill-defined “overnight culture,” technique for colony picking, pellet lyophilization, amount and composition of gas headspace in culture vessels, type of flasks, shaking speed, optical density at harvest, lysis method, and use of clarification steps.
There are some open resources for information sharing, such as OpenWetWare.org. The U.S. National Institute of Standards and Technology (NIST) is engaged in investment and outputs, in close partnership with researchers using cell-free systems, to advance progress and to support economically-viable applications for bioengineering and biomanufacturing.
Source: NIST

