Inhibrx, Inc is a clinical-stage biotechnology company focused on developing a broad pipeline of novel biologic therapeutic candidates.
Inhibrx uses diverse methods of protein engineering to address the specific requirements of target and disease biology, including its proprietary sdAb platform. The Inhibrx pipeline is focused on oncology, orphan diseases and infectious diseases. The company has a collaboration with Celgene and has received awards from several granting agencies, including NIH, NIAID and CARB-X.
sdAb Platform
According to the company, sdAbs provides a small, simple, modular target binding domain that can be combined in a variety of ways to meet the unique needs of biological targets. Discovery, humanization and optimization of sdAbs and their incorporation into unique configurations enables therapeutic candidates with favorable drug-like properties and differentiated mechanisms of action. Inhibrx creates various multivalent and multispecific therapeutic formats that are each designed to achieve unique functions specifically tailored to the requirements of a given biological target, including the ability to:
- effectively cluster receptors with precisely defined valency;
- simultaneously engage multiple antigens or epitopes;
- combine synergistic functions in a single molecule; and
- restrict therapeutic activity to specific micro-environments in the body.
While the benefits of sdAb proteins are well documented, the presence of pre-existing anti-drug antibodies in human serum may limit the broad use of these agents as therapeutics. As such the company has developed a series of patented modifications to its humanized sdAb scaffold that are designed to eliminate recognition by these pre-existing anti-drug antibodies, without compromising affinity, specificity or stability of the sdAb. It believes this improvement, combined with its extensive knowledge of the structural nuances of sdAbs, will enable the streamlined development of therapeutic candidates with favorable biophysical properties, and will greatly extend the utility and applicability of its sdAb platform.
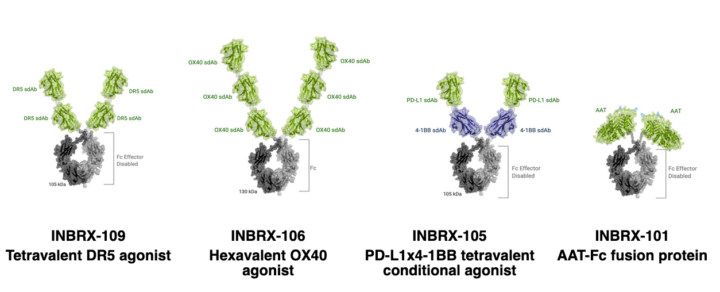
INBRX-109
The company’s most advanced therapeutic candidate, INBRX-109, is a tetravalent death receptor 5, or DR5, agonist being evaluated in patients diagnosed with chondrosarcoma and mesothelioma, two difficult-to-treat cancers. INBRX-106 is a hexavalent OX40 agonist currently being investigated in patients with locally advanced or metastatic solid tumors. Both INBRX-109 and INBRX-106 programs are designed to achieve target agonism through precise control of therapeutic valency. INBRX‑105 targets two proteins, each of which can lead to an enhanced anti-tumor immune response; it is engineered with the goal of improving safety through conditional target agonism. Based on preclinical mechanistic data, INBRX-106 and INBRX-105 may have the potential to be used in combination with, or in place of, currently approved checkpoint inhibitors. The company’s fourth program, INBRX-101, is an optimized, recombinant alpha 1 antitrypsin, or AAT, augmentation therapy for AATD. Inhibrx anticipates additional data releases from all four of our clinical programs by the end of 2021.
Bluebird Collaboration
In 2019, bluebird bio, Inc. and Inhibrx announced an exclusive license agreement to research, develop and commercialize chimeric antigen receptor (CAR) T cell therapies using Inhibrx’s proprietary single domain antibody (sdAb) platform to multiple cancer targets. The small size of sdAbs may enable the generation of more complex CAR T cell products such as those designed to combine additional functions into a single CAR molecule or recognize multiple tumor antigens simultaneously.
Financials
To date, the company has financed its operations through equity and debt financings, license and milestone revenue and grants. It has incurred significant recurring operating losses since inception. For the years ended December 31, 2018 and 2019, its net loss was $31.0 million and $51.4 million, respectively. Its net loss for the six months ended June 30, 2020 was $38.0 million. As of December 31, 2019 and June 30, 2020, it had an accumulated deficit of $69.3 million and $107.2 million, respectively.
As of June 30, 2020, we had $5.6 million in cash.
The ideas presented on this site do not constitute a recommendation to buy or sell any security. Investors are advised to conduct their own independent research into individual stocks before making a purchase decision. In addition, investors are advised that past stock performance is not indicative of future price action. You should be aware of the risks involved in stock investing, and you use the material contained herein at your own risk. Neither SYNTHETIC.COM nor any of its contributors are responsible for any errors or omissions which may have occurred. The analysis, ratings, and/or recommendations made on this site do not provide, imply, or otherwise constitute a guarantee of performance. SYNTHETIC.COM posts may contain financial reports and economic analysis that embody a unique view of trends and opportunities. Accuracy and completeness cannot be guaranteed. Investors should be aware of the risks involved in stock investments and the possibility of financial loss. It should not be assumed that future results will be profitable or will equal past performance, real, indicated or implied. The material on this website is provided for information purpose only. SYNTHETIC.COM does not accept liability for your use of the website. The website is provided on an “as is” and “as available” basis, without any representations, warranties or conditions of any kind.

