This U.S. Military Lab Explores COVID-19’s Genetic Code
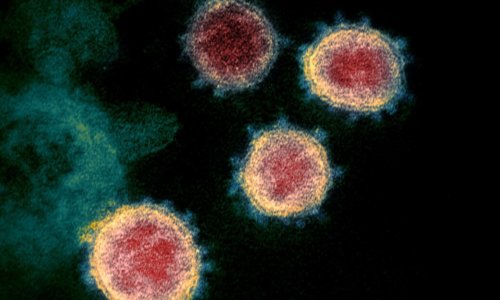
The U.S. military believes the key to finding a vaccine or effective treatment against COVID-19 may lie in its genetic code. Researchers around the globe are searching for clues to unlock the secrets of the novel coronavirus as the epidemic continues to spread worldwide. The Air Force Genetics Center of Excellence at Keesler Air Force … Read more